Highly efficient, stable and low Pt loading oxygen reduction catalyst is the main research direction of cathode materials for fuel cells, where core-shell Pt-based nanomaterials are of great significance.However, the structural instability of the ultrathin shells and the leaching of the core components make it difficult to meet the demands of stable battery operations. In regard to this, Yi Ding Research Group and Jun Luo Research Group, Tianjin University of Technology, and Limin Liu Research Group, Beijing Computational Science Research Center published a paper in Nature Energy, reporting a high activity, high stability, low Pt loading Pt-Pd-Au ternary alloy oxygen reduction catalyst. (https://www.nature.com/articles/nenergy2017111)
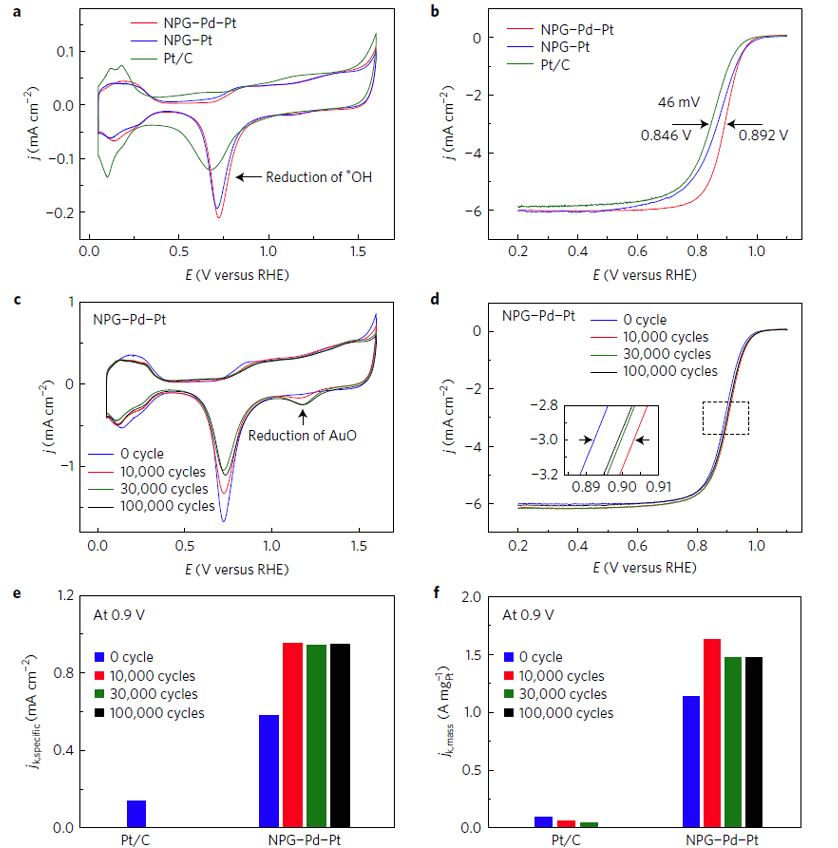
Figure 1. Electrochemical properties of nanoporous Au-Pd-Pt (NPG-Pd-Pt ) and samples after 10,000, 30,000 and 100,000 cycles.
Due to the differences in the atomic diffusion rates and electronegativity of Au, Pd and Pt, researchers applied electrochemical cyclic voltammetry to the surface structure of the Pt-Pd-Au shell on the sub-nanometer scale, and eventually obtained a high activity and stability Pt-Pd-Au ternary alloy core-shell oxygen reduction catalyst.The mass activity of the catalyst was 15 times higher than that of commercial Pt/C catalyst and remained unchanged after 70,000 cycles of stability test. The research findings were published in Nature Energy in August 2017. The paper was sponsored by the National 973 Program Project of China, the National Natural Science Foundation of China, the National Program for Thousand Young Talents of China.

Figure 2. High resolution HAADF and EDS-Mapping images of NPG-Pd-Pt catalyst

Figure 3. High resolution HAADF images and atomic-level EDS-Mapping images after 10,000 cycles (a-e) and 30,000 cycles (f-j) of NPG-Pd-Pt
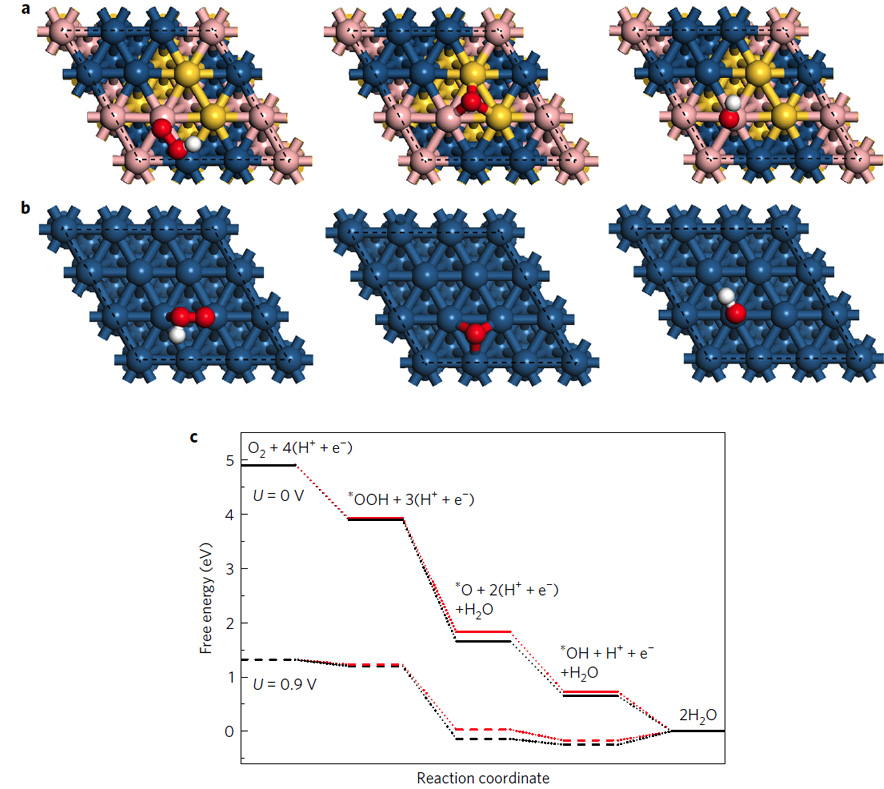
Figure 4. Theoretical calculation of ORR activity on Pt-Pd-Au(111) and Pt(111) surfaces

